Safety officials believe the bottles may pose a risk if swallowed by children, as they lack required safety features and warning labels.
More than 780,000 travel-size nasal spray bottles are being recalled because their packaging does not meet federal child safety requirements, Consumer Product Safety officials said.
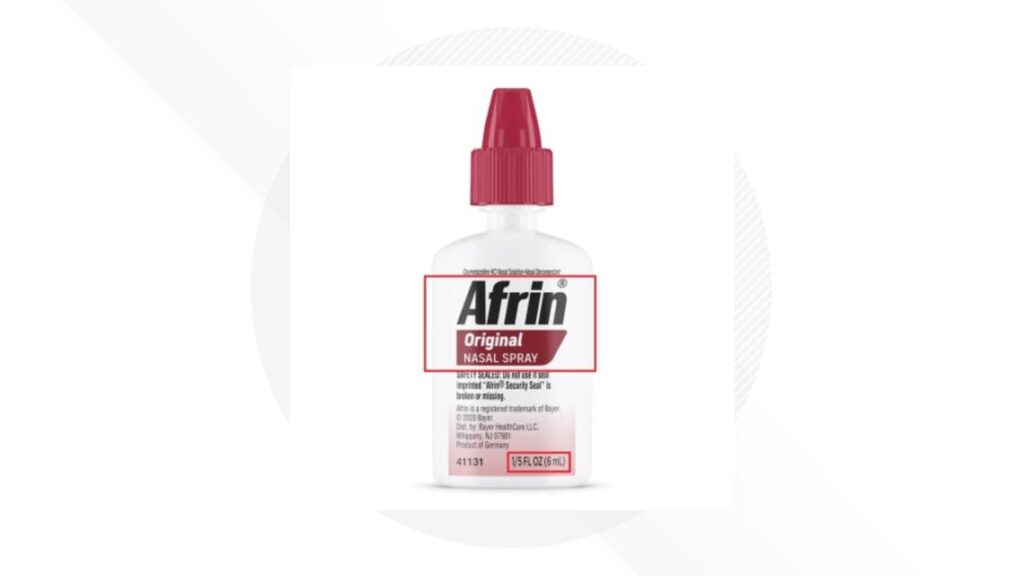
The recall involves about 786,100 bottles of Afrin Original Nasal Spray 6 mL distributed by Bayer HealthCare LLC.
According to the recall notice issued April 30, the 6 mL bottles contain an imidazoline, a type of medication that must be sold in child-resistant packaging under the Poison Prevention Packaging Act.
The affected products lack child-resistant features and required warning labeling, posing a risk of serious illness or injury if swallowed by young children.
No injuries or incidents have been reported.
The recalled products include unexpired travel-size bottles labeled “Afrin Original Nasal Spray” and “1/5 FL OZ (6 mL),” with specific lot numbers listed by the company. No other Afrin products or sizes are included in the recall.
The sprays were sold at convenience stores and travel hubs, including airports, between September 2024 and April 2026 for about $7 to $9.
Consumers are advised to keep the bottles out of sight and reach of children and request a refund through the company’s recall website. Customers will be asked to submit a photo of the product before disposing of it.
For more information, consumers can contact Bayer or visit its recall webpage.